Phenomenon Of Osmosis:
Osmosis is the most important physical process that brings about the movement of water in living systems as well as in their environment.
Osmosis is the phenomenon of diffusion of water or solvent from the region of its higher chemical potential to the region of its lower chemical potential when diffusion of solute is prevented by means of a semipermeable membrane.
It may also be defined as the net diffusion of water molecules from a dilute solution to the concentrated solution when the two are separated by means of a semi-permeable membrane. If the solution is separated from its pure solvent, the molecules of the solvent move from pure solvent to the solution.
When the water moves out of the cell due to hypertonic solution (of higher concentration), the type of osmosis is termed exosmosis. If the water enters the cell due to hypotonic solution (of lower concentration), the process is called endosmosis.
The process of osmosis can be easily demonstrated by an osmometer. For this, take a small thistle funnel and tie its mouth with a natural membrane taken from the bladder of sheep, pig or goat. Fill the funnel partially with a concentrated sugar solution having a drop of safranin stain. Mark the level of sugar solution on the stem of the funnel. Immerse the bulb portion of thistle funnel in a beaker containing water. After an hour or so, you will observe a rise in the level as a result of an increase in the volume of water inside the thistle funnel. The movement of water molecules through a membrane towards an area of greater concentration of a dissolved substance is known as endosmosis.
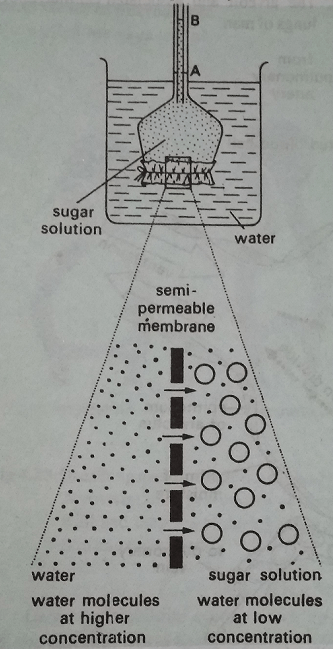
To understand why endosmosis happens, look at fig. The sugar molecules are larger than the water molecules. The natural membrane is selectively permeable or semipermeable containing tiny holes in it which are large enough to let the small water molecules through, but too small to let the larger sugar molecules through. Now the presence of the sugar molecules in the thistle funnel indicates that there is not much room for water molecules. So the water molecules inside the thistle funnel are less concentrated than the beaker outside. As a result, water molecules diffuse into the thistle funnel.
The inward movement of water due to endosmosis results in a pressure being built up inside the thistle funnel. We call this osmotic pressure. Osmotic pressure is the pressure developed by a solution when water moves into it across a semipermeable membrane. It is directly proportional to the difference in concentration to water molecules on either side of the membrane.
What is Osmosis Pressure?
Osmosis pressure of a solution is equivalent to the pressure which must be exerted upon it to prevent the flow of solvent into it across a semipermeable membrane. In other words, the maximum amount of pressure that can be developed in a solution separated from pure water by a semipermeable membrane is termed as osmotic pressure. It is measure in pascals, pa (1pa = 1 newton/m2). The osmotic pressure of a solution largely depends upon the ratio between the number of solute and solvent particles in a given solution. For example- a molar solution of sucrose has an osmotic pressure of approximately 22.4 atmospheres at 00C. However, a molar solution of sodium chloride has almost twice the osmotic pressure of a molar solution of sucrose. This is because sodium chloride is an electrolyte and dissociates almost completely into Na+ and Cl– ions (giving twice the particle number) whereas sucrose or glucose molecules are non-electrolyte and do not dissociate in water. The osmotic pressure of a given solution can be calculated by using the following relationship. Osmotic pressure = CST (where C is the molar concentration of a solution, S is the solution constant, which is 0.082 and T is the absolute temperature i.e. 0C + 273.









Comments (No)