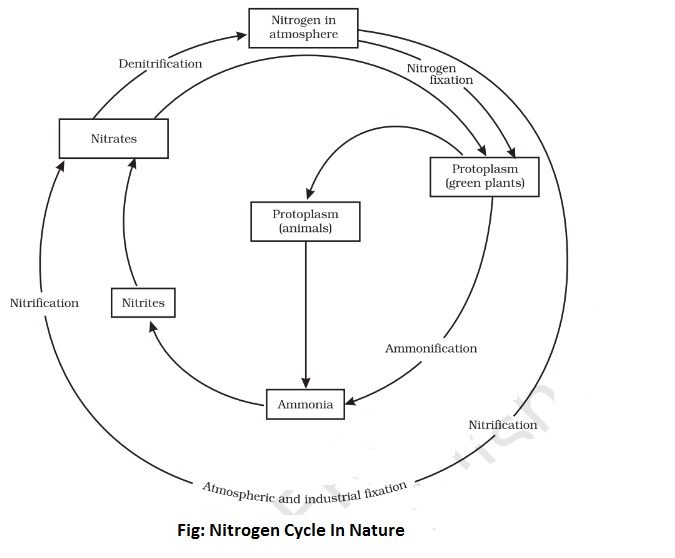
Working of Nitrogen Cycle in Nature:
Nitrogen is an essential constituent of all living organisms, namely, plants and animals. The atmosphere is the chief source of nitrogen. It is estimated that the atmosphere alone contains four thousand billion tonnes of nitrogen. Only a small quantity of nitrogen is present in the combined state and it is, thus, largely found in the free state.
The nitrogen of the air cannot be assimilated directly by the plants and animals. A few plants such as peas, beans and clove can, however, take the atmospheric nitrogen directly. Other plants generally assimilate nitrogen in the form of soluble compounds of nitrogen present in the soil.
On the other hand, animals derive their nitrogen from such substances like egg, meat, milk, peas, beans and other products of plants. As a consequence of this, the nitrogen is being continuously consumed from the atmosphere by the operations of various artificial and natural processes. In spite of all this, the overall percentage of nitrogen in the atmosphere more or less remains the same. This is due to the fact that the combined nitrogen is constantly passing through various processes back to the atmosphere.
The cycle of changes involving the conversion of atmospheric nitrogen into its compounds and their subsequent decomposition back into the nitrogen is referred to as nitrogen cycle. This process of nitrogen ever continues in nature. As a result, the percentage of nitrogen in the atmosphere always remains the same. The entire journey of nitrogen from atmosphere to soil and then to plants and animals and finally back to the atmosphere mainly involves the following stages-
(1) From air to soil- The nitrogen of air is converted into nitrogenous compounds which are subsequently mixed with soil.
(a) By lightning- On a rainy day, nitrogen and oxygen of air combine under the influence of electric discharge in the sky to give nitric oxide (NO). It combines with more oxygen to form nitrogen dioxide (NO2). This dioxide reacts with water of rain and more oxygen to give rise to the formation of nitric acid (HNO3).
N2 + O2 ————–> 2NO
2NO + O2 ——————> 2NO2
4NO2 + O2 + 2H2O ————-> 4HNO3
Nitric acid so formed goes to the soil in the form of rain. Then it reacts with calcium carbonate (CaCO3) present in the soil to yield calcium nitrate Ca(NO3)2 which serves as a food for plants.
(b) By using artificial manures- Atmospheric nitrogen is constantly removed by its conversion to ammonia, urea, ammonium sulphate and calcium ammonium nitrate. These compounds are good nitrogen fertilizers which are added to the soil to maintain its fertility and thus serve as a food for the growth of plants.
(2) From air to plants- Certain plants of Leguminous variety such as peas, beans, gram, clove etc. take up nitrogen directly from the air for their growth. These plants have small nodules on their roots. In these nodules, they have symbiotic bacteria which take up directly the atmospheric nitrogen and convert it into nitrogenous compounds in the soil. These compounds serve as food for the growth of plants.
(3) From soil to plants- Certain other plants assimilate nitrogen from nitrogen compounds like ammonium salts, nitrites and nitrates present in the soil. These plants convert the nitrogen present in these compounds into plant proteins under the influence of sunlight and chlorophyll (green colouring matter of plants).
(4) From plants to animals- Animals eat plants and hence proteins in the form of food. In the body, the proteins are decomposed into urea and amino-acids which are either converted into body proteins or excreted in the form of waste products.
(5) From plants and animals back to soil and air- The daily excretions or waste products of animals undergo decomposition under the influence of various bacteria such as ammonifying bacteria, nitrosifying bacteria and nitrifying bacteria into the corresponding ammonium compounds. Similarly, the death and decay of plants and animals also results in the formation of ammonium compounds.
Some ammonium compounds so formed are converted into free nitrogen under the influence of denitrifying bacteria. This free nitrogen goes into the air in order to make up the deficiency of nitrogen created by plants and animals.
As a consequence, of all these processes, a balance is maintained and nitrogen neither accumulates nor depleted. In nature, the nitrogen cycle is continuously repeated.
- Define Photosynthesis. Write down the importance of Photosynthesis
- What Are Plant Auxins? Discuss the role of Auxins (IAA) in the plant body?
- What Are Gibberellins? Discuss the functions of Gibberellins in the plant body?
- What Are Cytokinins? Discuss the functions of Cytokinins in the plant body?
- Air Pollution- Causes, Effects, and Control Measures
- Soil Pollution- Sources, Effects and Control Measures
- Tamil Board: Living World









Comments (No)