Table of Contents
Coordinate Covalent Bond:
A Coordinate Covalent Bond is a special type of covalent linkage in which both the electrons for sharing between two atoms are contributed by one of the participating atoms only and this pair of electrons is counted towards the stability of both the atoms. Thus, a coordinate covalent bond always occurs in association with an electrovalent or covalent bond or bonds in the same molecule. This bond is also sometimes referred to as a dative bond. The compounds formed by the coordinate linkage are referred to as coordinate compounds.
We shall now discuss some typical examples to illustrate the formation of the coordinate covalent or simply coordinate bond-
- Formation of Ozone- A molecule of ozone is formed when one of two oxygen atoms contained in an oxygen molecule contributes a lone pair of electrons to a third oxygen atom.
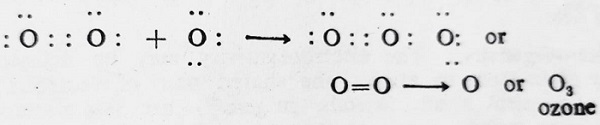
- Formation of Ammonium ion (NH4+)- In ammonia, the nitrogen atom has a lone pair of electrons which is donated to a hydrogen ion (proton) in order to give it a stable electronic configuration of inert gas helium and the donor nitrogen atom thus attains the stable electronic configuration of an inert gas neon. This gives rise to the formation of a coordinate bond. It is expressed as-
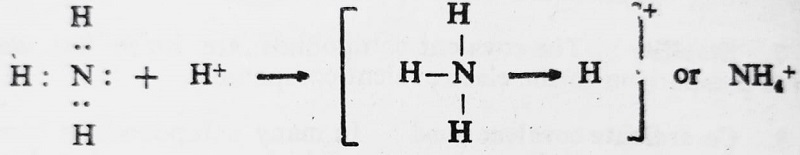
Characteristics of Coordinate Compounds:
The coordinate compounds show the following important characteristics-
- Nature- The coordinate compounds are molecular in nature i.e. they consist of molecules held together by weak forces of attraction.
- Melting and boiling points- The melting and boiling points of coordinate compounds are usually higher than those of the corresponding covalent compounds and lower than those of electrovalent compounds.
- Solubility- Like covalent compounds, the coordinate compounds are usually insoluble in water but are soluble in non-polar solvents like benzene, ether, acetone etc.
- Non-ionic nature- Like covalent compounds, these compounds do not form ions when dissolved in solvents like benzene, toluene, ether etc.
- Electrical conductivity- In solution or in fusion state, the coordinate compounds are poor conductors of electricity In polarity, they are more polar than the corresponding covalent compounds and less polar than the electrovalent compounds.
- Space Isomerism- Like a covalent bond, the coordinate covalent linkage is rigid and directional. Different space models of single coordinate compounds are possible. Thus, they are capable of showing the phenomenon of space isomerism.
- Stability- Like covalent compounds, the coordinate compounds are less stable towards heat as they are made up of molecules that are held together by the weak forces of attraction which can be easily overcome by heat. Hence, they are less stable towards heat.
Electronegativity:
Electronegativity may be defined as the tendency of an atom to attract the shared pair of electrons towards itself in a covalent bond. Metals, in general, are less electronegative than non-metals. Small atoms attract electrons more strongly than large atoms. In other words, atoms with small sizes are more electronegative in nature.
The elements having nearly filled electronic shells such as halogens are more electronegative than those which have fewer electrons in their valence shell. Different elements have different values of electronegativity.
The electronegativity values of some elements are as under-

| What are Polar and Non-polar Covalent Bonds? | Brownian Movement and Tyndall Effect |
| Graham Law of Diffusion | Ohms Law |
| Heat and Work | Huygens Principle |
| Heat and Temperature | Force and Motion– Tamil Board |









Comments (No)