Experimental Measurement of Osmotic Pressure:
Berkeley and Hartley’s method is the most commonly used method to determine the osmotic pressure of the solution. The apparatus used in this method is shown below.
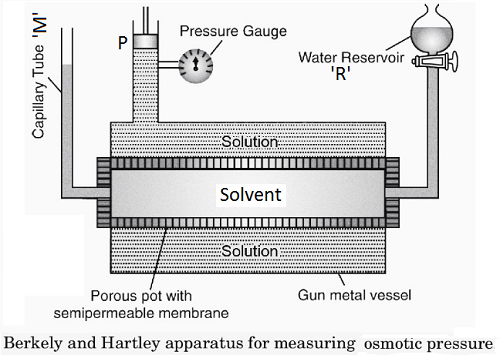
The apparatus consists of a porous tube containing the semi-permeable membrane of copper Ferrocyanide. One end of it is fitted with reservoir ‘R’ and the other end with capillary tube ‘M’ to indicate the level of solvent. The tube is filled with solvent. This tube is fitted into an outer vessel made of gunmetal and is fitted with piston ‘P’ and pressure gauge. It is filled with a solution whose osmotic pressure is to be determined. As soon as osmosis starts, the level of solution increases and piston ‘P’ moves up. Then additional weights are placed on the piston, which is sufficient to stop the osmosis and the level of the solvent remains steady in capillary tube ‘M’. The pressure, then, indicated by the pressure gauge is the measure of the Osmotic pressure of the solution.









Comments (No)