Table of Contents
Nuclear Fission and Nuclear Fusion:
Nuclear Fission:
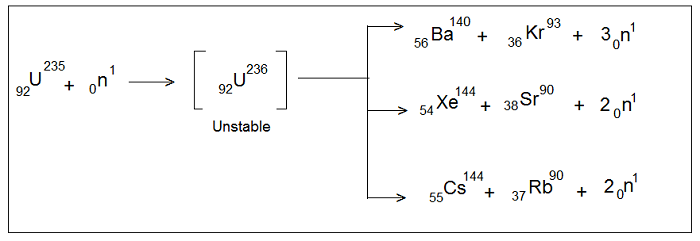
When a nucleus is bombarded with some sub-atomic particles (like α-particles, neutrons, protons etc.), the mass of the daughter element formed is either slightly greater or smaller than the parent element. However, it was observed by Hahn and Strassmann (1939) that when U-235 is bombarded by slow neutrons, U-236 is produced which being unstable split up into a number of fragments each of which has a mass much smaller than Uranium.
“The process of splitting of a heavier nucleus like that of U-235 into a number of fragments of much smaller mass by bombardment with slow-moving projectiles liberating a quite large amount of energy is known as Nuclear Fission”. For example- in the nuclear fission of U-235, the two main fragments are Ba and Kr along with the ejection of 2 or 3 neutrons and a huge amount of energy.
The neutrons released from the fission of the first ‘U’ atom hit other ‘U’ nuclei and cause their fission resulting in the release of more neutrons which further continue the fission process. In this way, a nuclear chain reaction is set up releasing a huge amount of energy. The amount of energy released is very high even though some of the released neutrons escape unused from the surface. The amount of energy available from 1 Kg of U-235 is equivalent to that available from 20,000 Kg of coal.
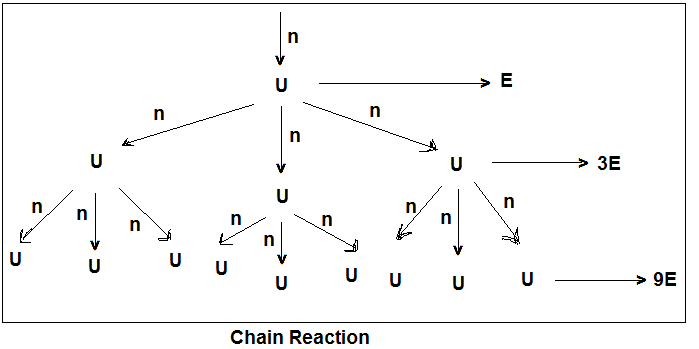
For the nuclear chain reaction to continue, there must be sufficient fissionable material. The minimum amount that the fissionable material must have so as to continue the chain reaction is called critical mass. For U-235, it lies between 1-100 Kg. Natural Uranium contains mostly U-238 (99.3%) which is not fissionable with slow neutrons. Another practically important nuclear fission is that of Pu-239. The energy released for every ‘U‘ atom undergoing fission is about 261.77 MeV (1MeV = 1.602 X 10-13 J). The tremendous amount of energy released during Nuclear Fission is because of the loss in mass which gets converted into energy according to Einstein’s equation i.e. E = mc2.
Nuclear Fusion:
It is just opposite to Nuclear Fission and is defined as “the process in which lighter nuclei fuse together to form a heavier nucleus with the liberation of a tremendous amount of energy”. The release of energy is due to loss in mass i.e mass defect. These reactions take place at higher temperature (generally greater than 106 K) in order to overcome the electrostatic repulsion between the fusing nuclei and so these reactions are also known as Thermonuclear Reactions. In nature, such a high temperature is available only in the Sun which contains about 73% Hydrogen, 26% Helium and 1% other elements. It is, therefore, believed that in the Sun, the following fusion process takes place-
| 4 1H1 ————–> 2He4 + 2 +1e0 + γ |
Calculations show that this process is accompanied by the evolution of about 26 MeV of energy which is available to us from the Sun and which keeps the Sun at extremely high temperature.









Comments (No)