Table of Contents
What is Electrochemical Cell?
- The electrochemical cell is the device that is used to convert the chemical energy produced in a redox reaction into electrical energy.
- It is also called Galvanic Cell or Voltaic Cell after the names of Luigi Galvani (1780) and Alessandro Volta (1800) who were the first to perform experiments on the conversion of chemical energy into electrical energy.
Construction and Working of Electrochemical Cell:
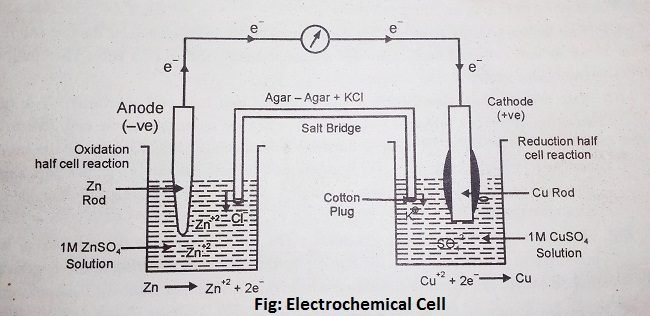
In an electrochemical cell, there are two separate containers. In one container Zinc rod is dipped into the 1M ZnSO4 solution and another container contains a copper rod that is dipped into the 1M CuSO4 solution. In the first container, Zinc acts as an anode, so oxidation takes place, Zn liberates electrons and converted into Zn2+ i.e.
Zn ————————> Zn2+ + 2e– (Anode)
While in the second container copper acts as the cathode, so reduction takes place. Copper accepts electrons that are liberated by the Zn rod. The reaction is as follows-
Cu+2 + 2e– ———————–> Cu (Cathode)
The electrons released by the oxidation of Zn to Zn2+ ions will be accepted by the Cu+2 ions of CuSO4 in the other half cell and the latter will be reduced to copper. The positively charged Zn2+ ions pass into the solution. After sometimes, this results in the accumulation of extra positive charge in the solution around the anode. Similarly, due to the reduction of some Cu+2 ions to Cu, the solution around the cathode will acquire extra negative charge due to excess of SO42- ions. The accumulation of positive charge around the zinc rod takes place. Similarly, the accumulation of negative charge around the copper electrode will prevent the flow of electrons to the copper ions. Thus, the flow of electrons will occur only momentarily and the cell will stop working.
The two containers involving oxidation and reduction half-reactions are called half cells. The zinc rod dipped into a ZnSO4solution is oxidation half cell and the copper electrode dipped into a CuSO4 solution is reduction half cell.
Functions of Salt Bridge in the Electrochemical Cell:
Salt Bridge is a U-shaped tube that is filled with saturated KCL and agar-agar solution, then both the ends of the salt bridge are closed with a cotton plug and put this salt bridge into the two containers of the electrochemical cell.
The important functions of the salt bridge are:
- Salt bridge complete the electrical circuit: The salt bridge connects the two solutions of the half cells and their electrodes are connected by means of a wire. Therefore, the salt bridge completes the circuit.
- Salt bridge maintains the electrical neutrality of two half-cell solutions: The main function of the salt bridge is to provide the electrical neutrality of the cell. Due to the accumulation of Zn2+ ions into the first container and SO42- ions into the second container, the cell stops working. Just to restart the cell, the salt bridge provides anions into the first container to neutralize the Zn2+ and cations to the second container to neutralize the SO42-.
- Eliminates Liquid Junction Potential: The third function of the salt bridge is to minimize or almost eliminate the liquid junction potential occurring at the interface of the electrolyte salt solution and the ends of the salt bridge due to the diffusion of ions.









Comments (No)