Table of Contents
Vapour Pressure:
It is the pressure exerted by vapours of liquid when the liquid and vapours are in equilibrium with each other in a closed system. It is the characteristic of each liquid and it changes with temperature.
Factors affecting the vapour pressure of a liquid:
(1) Nature of Liquid- Weaker are the intermolecular forces, greater is the amount of vapours because of more tendency of liquid to pass into the vapour phase and vice-versa. So, liquids with low intermolecular forces have generally high vapour pressure.
(2) Temperature- Higher the temperature, the greater is the vapour pressure. It is due to the reason that increased temperature; increases the Kinetic energy of molecules and hence molecules can leave the liquid and pass into the vapour phase.
(3) Nature of solute added to the solvent to get solution- The volatile solute does not alter the vapour pressure of liquid solvent at a particular temperature. Whereas non-volatile solute decreases the vapour pressure of the solvent (liquid) because such solute has no escaping tendency to pass into the vapour phase.
However, the effect of the nature of solute on the vapour pressure of the solution is given in terms of Raoult’s law.
Raoult’s Law:
In 1887, Raoult put forward a law which gives the relationship between the mole fraction of solute (volatile as well as non-volatile) and its vapour pressure in a solution called Raoult’s Law.
Raoult’s Law for Volatile Solutes:
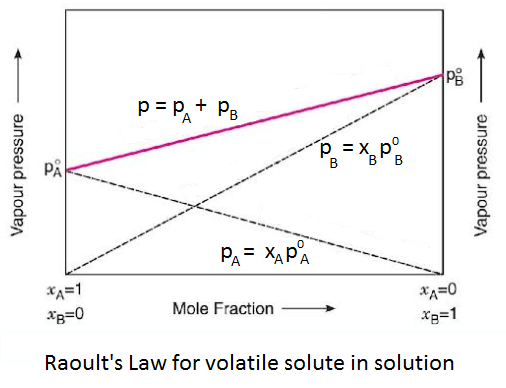
It states that “in a solution of volatile solutes, the partial vapour pressure of each component of solution is directly proportional to its mole fraction in solution”.
Let us consider a binary mixture of two miscible volatile liquids A and B, having mole-fraction xA and xB respectively. Suppose at a certain temperature, their partial pressures are pA and pB, respectively.
| According to Raoult’s Law, pA ∝ xA and pB ∝ xB or pA = p°A . xA and pB = p°B . xB |
Where p°A and p°B are the vapour pressure of components A and B in pure states. Thus, the total pressure (P) is given by Dalton’s law, i.e.,
| p = pA + pB = p°A . xA + p°B . xB |
Raoult’s Law for Non-Volatile Solutes:
According to Raoult’s Law for a binary solution of two components (both volatile and miscible) is-
| p = pA + pB ⇒ P = p°A . xA + p°B . xB ……….(i) |
Let ‘A’ represent solvent and ‘B’ represent solute.
If the solute is Non-Volatile (for example- Glucose, Urea), it will not contribute to the total vapour pressure of the solution, therefore, p°B = 0.
| Thus, p = pA = p°A . xA ……….(ii) But we know that for a binary solution, xA + xB = 1 ⇒ xA = 1 – xB ……….(iii) |
Using relation (iii) in relation (ii), we get
| pA = p°A (1 – xB) = p°A – p°A . xB ⇒ pA = p°A – p°A . xB ⇒ p°A . xB = p°A – pA ⇒ xB = (p°A – pA)/p°A |
In this expression (p°A – pA) is the lowering of vapour pressure and (p°A – pA)/p°A is the relative lowering of vapour pressure. This expression is the mathematical form of Raoult’s Law for non-volatile solutes and is expressed as-
“The relative lowering of the vapour pressure of a solution containing non-volatile solute is equal to the mole fraction of solute in the solution”.









Comments (No)