Structure of the Candle Flame:
Candle wax is a mixture of solid carbon compounds called hydrocarbons. When a candle is lighted, the wax melts, rises up the wick and gets converted into vapour. These wax vapours burn in the air producing a yellow, luminous flame. Like a kerosene lamp, the candle flame is also yellow and luminous. The candle flame is yellow and luminous because of the presence of unburnt carbon particles in it due to the incomplete combustion of wax hydrocarbons in insufficient supply of oxygen (of air) for burning. The incomplete combustion of wax hydrocarbons produces some unburnt carbon particles. These small carbon particles rise in the flame, get heated and start glowing (giving out light) of yellow colour. These carbon particles then leave the flame as smoke and soot.
A candle flame actually consists of three zones:
The three zones of a candle flame have different colours. The innermost zone is black, the central zone or middle zone is yellow whereas the outer zone is blue in colour.
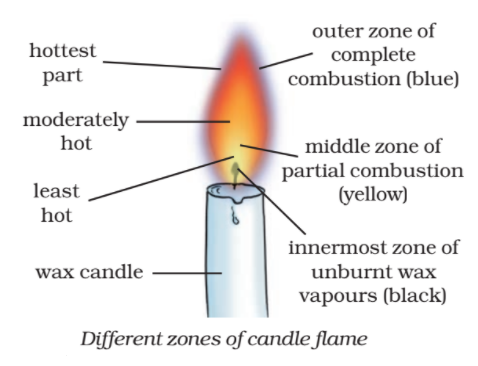
- The innermost zone of a candle flame is dark or black. This is due to the presence of the unburnt vapours of the wax. This innermost zone is the least hot part of the candle flame.
- The central zone or middle zone of the candle flame is yellow, bright and luminous. The fuel vapours or wax vapours burn partially (incompletely) in this zone because there is not enough oxygen (or air) for burning in this zone. This incomplete or partial burning of the wax produces some carbon particles. These carbon particles become white-hot (or rather yellow-hot) and emit yellowish light. So, it is the glow of unburnt carbon particles which makes the middle zone luminous (light-giving). This middle zone of the candle flame produces a moderate temperature. This zone is the major part of the candle flame.
- The outer zone of the candle flame is blue, non-luminous zone. In this zone, complete combustion of wax vapours takes place because there is plenty of air around it. The outermost zone has the highest temperature in the flame.









Comments (No)