Table of Contents
Dialysis and Electro-Dialysis:
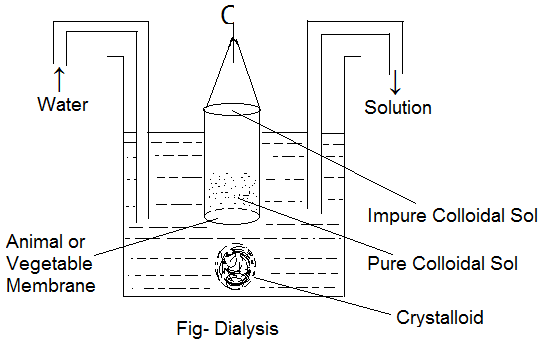
Dialysis:
The process of separation of a colloid from a crystalloid or electrolyte by making use of animal or vegetable membrane (semi-permeable membrane) is known as Dialysis. It is based upon the fact that crystalloid particles because of their small particle size can slowly diffuse through the pores of the semipermeable membrane but colloidal particles because of their larger size can not do so. The process is carried out in a Dialyser which consists of a hollow cylinder at the lower end of which an animal or vegetable membrane is tied down. The impure colloidal sol is placed in the dialyser which is suspended in an outer vessel containing water. The small particles or ions of the crystalloid or electrolyte present as an impurity in colloidal sol diffuse out of the membrane while the bigger colloidal particles are retained in the dialyser. Water is continuously recirculated in order to increase the rate of dialysis.
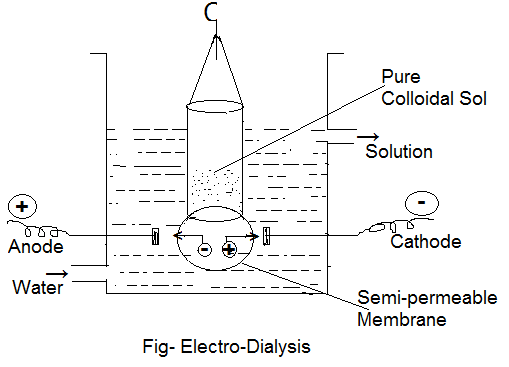
Electro-Dialysis:
Dialysis is a slow process and may take days for completion. The rate of dialysis can be accelerated by making use of electrodes across the membrane containing impure colloidal sol. This process is known as electrodialysis and is defined as the process of the separation of electrolyte particles from a colloidal sol using a semipermeable membrane under the influence of an applied electric field. In this process, an electric field is set up by putting electrodes in the tank containing Dialyser. The positively and negatively charged ions of the electrolyte (present as an impurity in colloidal solution) move towards the oppositely charged electrodes through the animal or vegetable membrane of the dialyser. The colloidal sol in the purified state is retained in the dialyser. This process cannot be used in case colloidal sol contains non-electrolytic impurity like urea, sugar, glucose etc. as these do not produce small ions in solution.









Comments (No)