Table of Contents
Emission and Absorption Spectrum:
Suppose EA and EB are the two possible states of an atomic or molecular system. The energy difference between the two states is given by Δe i.e.,
| ΔE = EB – EA |
Transitions can take place between two energy levels either by the absorption of energy or by the emission of energy by the system. The emitted or absorbed energy is in the form of electromagnetic radiations and energy can be expressed as,
| ΔE = hν Joules |
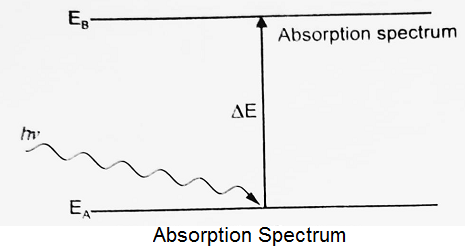
Absorption Spectrum:
When there is an absorption of radiations from EA to EB, the spectrum obtained will be the absorption spectrum. An absorption spectrum occurs when light passes through a cold, dilute gas and atoms or molecules in the gas absorb at characteristic frequencies. Since the re-emitted light is unlikely to be emitted in the same direction or same region of the electromagnetic spectrum as the absorbed photon, this gives rise to dark lines (absence of absorbed radiation) in the spectrum.
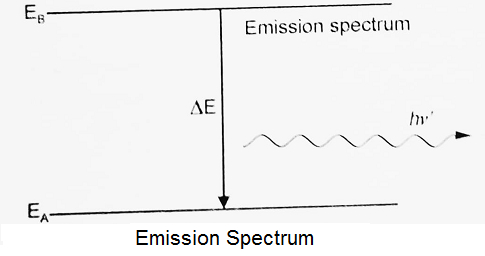
Emission Spectrum:
When there is the transition of an excited atom or molecule from a state of high energy (EB) to a state of lower energy (EA), the excess energy is emitted as a photon, the spectrum obtained will be emission spectrum. Thus emission lines correspond to photons of discrete energies that are emitted when excited atoms in the gas make transitions back to low lying energy levels.









Comments (No)