Table of Contents
What is Spectrum?
The ordered arrangement of radiations according to their wavelengths or frequencies is called Spectrum.
Origin of Spectrum:
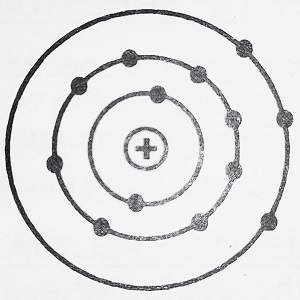
Every substance consists of atoms. Each atom has a positively charged central core called a nucleus. The nucleus contains protons and neutrons. The electrons revolve around the nucleus in various orbits as shown. The number of electrons in the orbits is equal to the number of protons in the nucleus. The electrons in the inner orbits have less amount of energy as compared to the energy of the electrons in outer orbits.
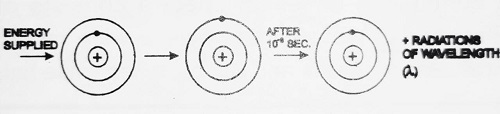
When external energy is supplied to the atom, the electron absorbs this energy and goes to the higher orbit. Now the atom is said to be in an excited state. The excited atom returns to its ground state after 10-8 sec, by emitting radiations of a certain wavelength as shown.
We know, From Bohr’s Postulate,
| hν = E2 – E1 ν = E2 – E1 / h C/λ = E2 – E1 / h (Where ν = C/λ) 1/ λ = E2 – E1 / Ch λ = Ch / E2 – E1 |
This is the required expression for a wavelength of emitted radiations.
Types of Spectra:
Spectra are classified into two categories-
- Emission Spectra.
- Absorption Spectra.
Emission Spectra:
An object on heating or passing electric discharge etc. gets excited to a higher energy state and starts giving out radiation. When this radiation is dispersed by a prism or any other device an emission spectrum is obtained. The emission spectrum gives valuable information about the source material. These are of three types-
- Continuous Emission Spectra.
- Line Emission Spectra.
- Band Emission Spectra.
Continuous Emission Spectra:
A continuous emission spectrum consists of unseparated wavelengths over a wide range. Incandescent solids and liquids, when white-hot, emit continuous emission spectra. Example- spectra of sunlight, an incandescent bulb etc. are continuous emission spectra. It depends on the temperature of the source.
Line Emission Spectra:
A line emission spectra consist of a few isolated bright coloured lines. It is emitted by the atoms in the gaseous state. For example- a sodium discharge lamp, a mercury vapour lamp etc. emit line emission spectra. It depends only on the nature of the substance emitting them.
Band Emission Spectra:
A band emission spectrum consists of a number of bright coloured bands. One end of each band is sharp and well defined. This end is called Band Head. The other end of the band, which is not so sharp is called the tail of the Band. A band emission spectrum is emitted by chemical compounds in a vapour state. When band emission spectra is examined with the help of a high power spectrometer, it is found that each band is further made up of a group of bright lines. For example- the spectra of iodine, oxygen molecules etc. is band emission spectra.
Absorption Spectra:
When a beam of light from a hot source is passed through a substance (at a lower temperature), a part of the light is transmitted but the rest of it is absorbed. With the help of a spectrometer, we can know the fraction of light absorbed corresponding to each wavelength. The distribution of the wavelength absorption of light by a substance is called an absorption spectrum. Every substance has its own characteristic absorption spectrum. The absorption spectra are of three types-
- Continuous Absorption Spectra.
- Line Absorption Spectra.
- Band Absorption Spectra.
Continuous Absorption Spectra:
When we place red glass in the path of rays, it absorbs all the colours up to red. Hence we get continuous absorption spectra up to red colour.
Line Absorption Spectra:
When light from the source is allowed to pass through gas or vapour, then this gas absorbs certain wavelengths and hence dark lines are produced. Then these dark lines are called line absorption spectra.
Band Absorption Spectra:
When white light is allowed to pass through a dilute solution of blood, then we observe band absorption spectra.









Comments (No)